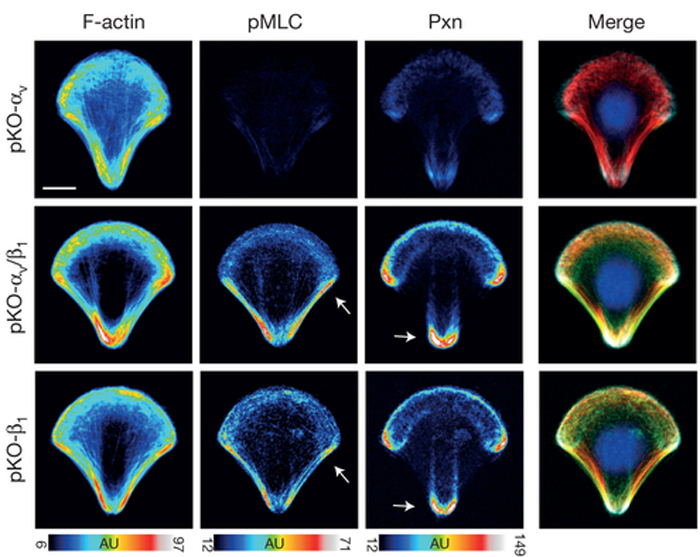
Integrins are transmembrane proteins that attach a cell to another cell or its surrounding extracellular matrix, so their role in regulating cell adhesion in normal cell function, development, and disease is of serious interest to biologists. There are several types of integrins, frequently expressed together at the same time. A recent paper describes a system to investigate how specific integrins function in cell adhesion, and found notable differences. Schiller and colleagues expressed either β1- or αv-class integrins in fibroblasts lacking all other integrins, and found that β1-class integrins are important in small peripheral adhesions, while αv-class integrins are important for large focal adhesions. In the images above, activity of the actin-based motor myosin and presence of the focal adhesion protein paxillin are shown in cells plated on micropatterned shapes. Cells with both β1- and αv-class integrins (middle row) have the highest myosin activity (pMLC) and paxillin signal, while cells with either β1- or αv-integrin alone have lower myosin activity and paxillin signal.
Adapted by permission from Macmillan Publishers Ltd, copyright ©2013
No comments:
Post a Comment